Participant Interactions

We are seeking volunteers comprising 40 ME/CFS patients and 20 controls to participate in this NIH funded (U54AI178855) project. The ME/CFS subjects will be identified by expert physician Dr. Susan Levine, M.D. (Manhattan, NY). Only those participants whose onset of ME/CFS was before 2020 will be eligible. Please review our recruitment flyer for more information on how to volunteer as a patient or control in this research study.
The volunteers will fill out a large number of online surveys that will characterize various aspects of their current and past health, as well as specific ME/CFS symptoms. In a one-time visit to the Hospital for Special Surgery, each participant will provide a blood and a urine sample and undergo a muscle biopsy. The muscle to be sampled will be the vastus lateralis, which is on the side of the thigh, part of the quadriceps muscle.
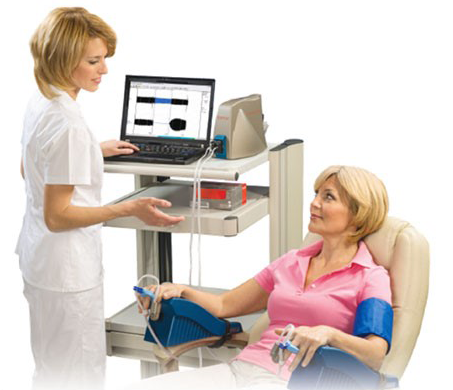 In addition, each subject will have their endothelial function examined with an EndoPat® device, which measures reactive hyperemia.
In addition, each subject will have their endothelial function examined with an EndoPat® device, which measures reactive hyperemia.
The subjects will also perform a hand-grip strength test.
By analyzing all the data together, it may be possible to correlate particular symptoms with molecular disruptions, endothelial dysfunction, and level of muscle strength/weakness.
Samples banked from our prior two-day exercise test experiments will also continue to be used for cell-free RNA assays, identifying extracellular vesicles cargo, and endothelial function studies.
Patient interactions will occur at the Hospital for Special Surgery (HSS, Manhattan, NY), under the direction of Dr. David Fernandez, and Dr. Yoshimi Endo, M.Ds.
Dr. Endo will acquire the muscle biopsies. Dr. Laura Donlin at HSS will oversee fractionation of blood and preparation of urine and muscle biopsy samples for shipment to Cornell-Ithaca.

 Center for Enervating NeuroImmune Disease
Center for Enervating NeuroImmune Disease